Key Takeaways
- Claritas Axis has introduced a 10-question Drug Supply Chain Readiness Snapshot, designed specifically for eye care settings such as retina, oculoplastics, and optometry practices
- A simple 10-question readiness snapshot can help practices quickly identify documentation gaps before they become compliance issues
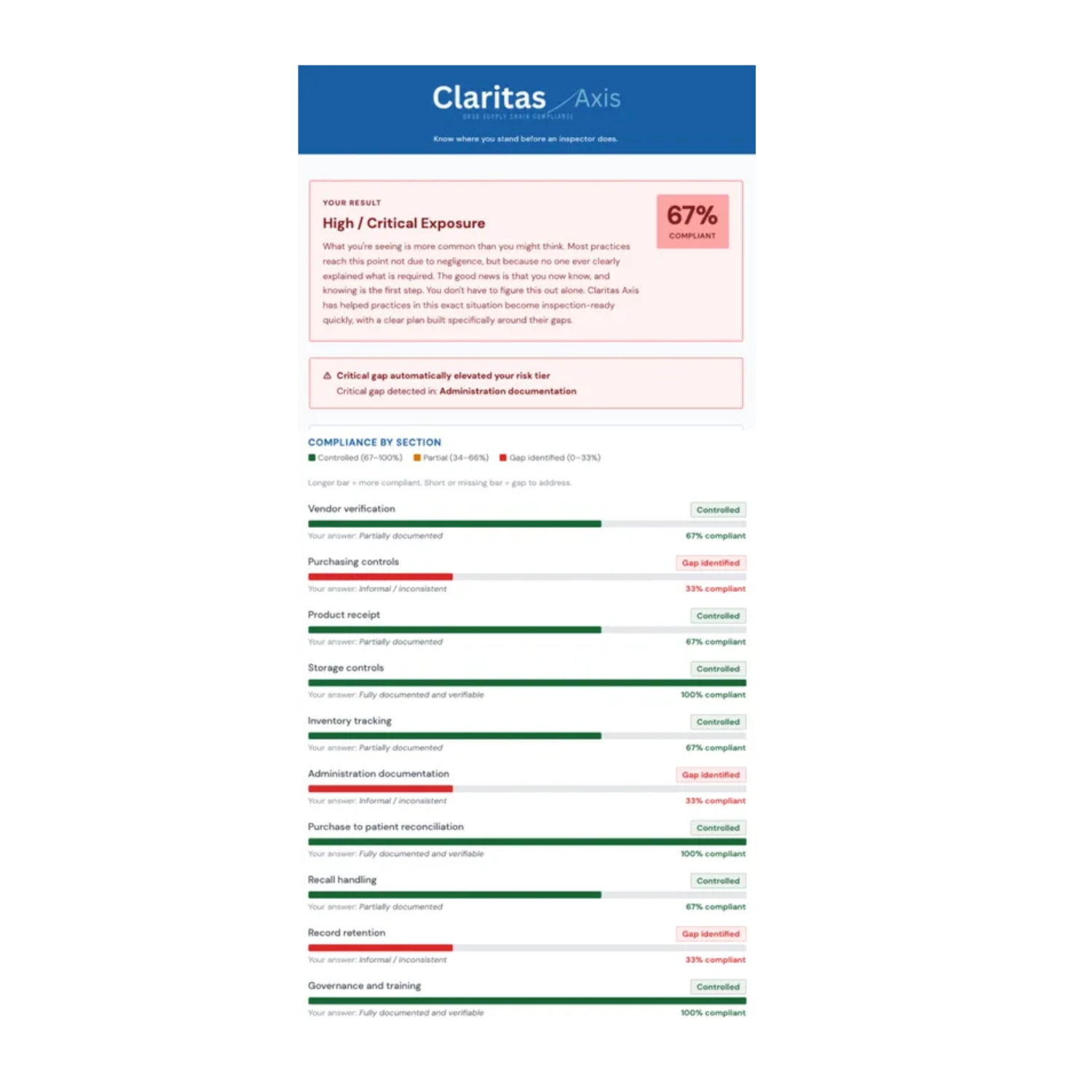
According to Claritas Axis, a growing compliance issue is quietly moving up the agenda for eye care providers: readiness for federal drug supply chain requirements. As enforcement of the Drug Supply Chain Security Act (DSCSA) continues to take hold, a new free assessment tool is aiming to help practices determine whether they are prepared—or potentially exposed.
Claritas Axis has introduced a 10-question Drug Supply Chain Readiness Snapshot, designed specifically for eye care settings such as retina, oculoplastics, and optometry practices. The tool is designed to offer a quick way to evaluate whether a practice’s drug documentation and procedures would stand up to inspection.
While DSCSA is often associated with pharmacies and large health systems, its scope extends to any “dispenser” that purchases and administers prescription drugs. That definition captures a wide swath of eyecare.
Retina specialists routinely administer anti-VEGF therapies. Oculoplastics and neuro-ophthalmology practices use Botox and other injectables. Dry eye clinics may rely on compounded treatments. Meanwhile, some comprehensive ophthalmology and optometry practices have expanded into therapeutic and aesthetic services, including peptide therapies and IV protocols sourced through compounding pharmacies or IV vendors. The result is a regulatory footprint that reaches far beyond what many practices assume—and increasingly overlaps with newer service lines being marketed to patients.
According to James Strafuss, founder of Claritas Axis, many practices remain unaware of how directly DSCSA applies to them. That confusion has contributed to a significant preparedness gap, even though full federal enforcement began in 2023.
“Practices often believe that working with trusted vendors and keeping invoices is enough,” Mr. Strafuss notes. “But DSCSA requires more structured documentation and clearly defined procedures.”
Claritas Axis emphasizes that many eyec are practices have never evaluated their drug workflows from an inspector’s perspective. DSCSA expectations include not just proof of purchase, but also documented processes for verifying suppliers, handling suspect products, and maintaining traceability records.
The newly released readiness snapshot is intended as a starting point. In just 10 questions, it helps practices identify whether obvious gaps exist—and whether a more comprehensive review may be necessary.