Key Takeaways
- Elegrobart significantly improved key symptoms of chronic thyroid eye disease in a phase 3 trial, with responder rates exceeding placebo
- The treatment was generally well tolerated and offers a more convenient at-home injection option compared to existing therapies.
- Viridian plans to seek FDA approval in early 2027
Viridian Therapeutics announced positive topline data from the REVEAL‑2 phase 3 clinical trial of elegrobart in patients with chronic thyroid eye disease (TED).
Elegrobart, a monoclonal antibody targeting the insulin-like growth factor-1 receptor (IGF-1R), is designed to be administered via a subcutaneous injection, potentially allowing patients to self-administer the treatment at home.
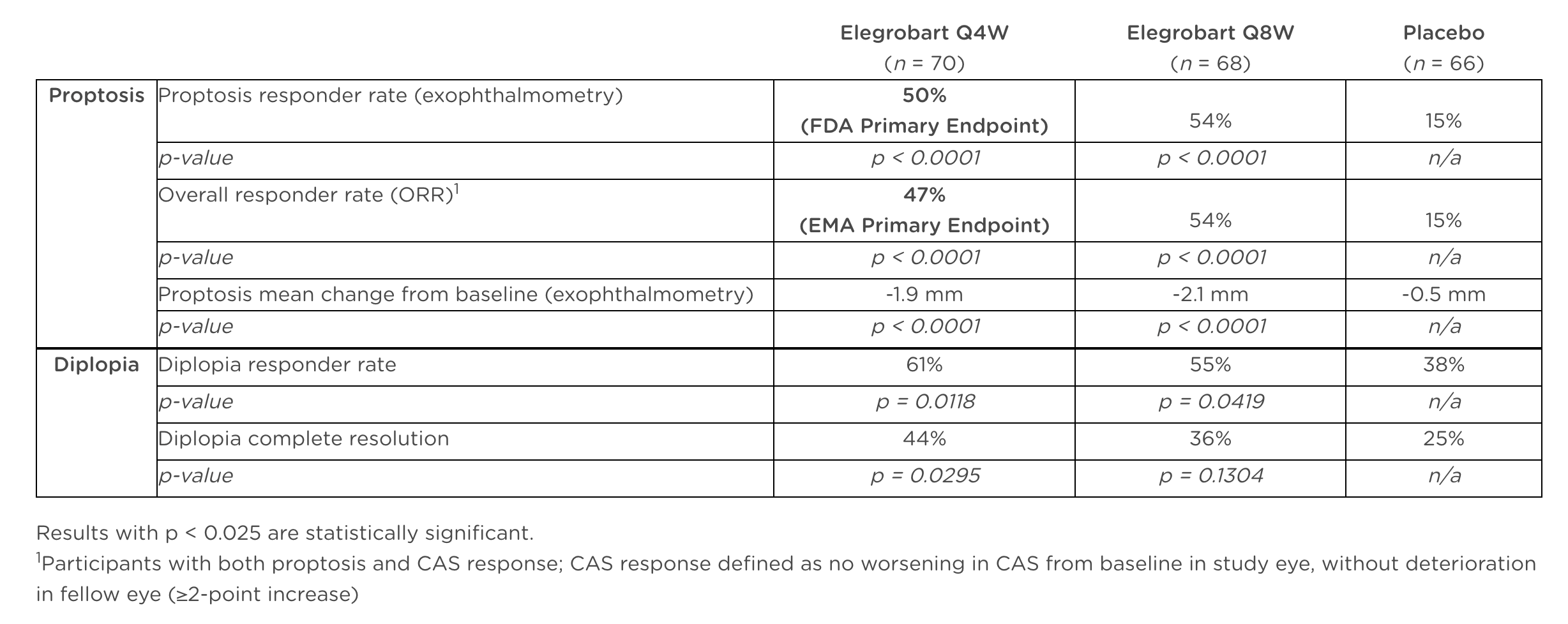
The REVEAL-2 trial enrolled 204 patients, testing two dosing schedules—every 4 weeks and every 8 weeks—against placebo. Both dosing regimens achieved high statistical significance in reducing proptosis, a hallmark symptom of TED characterized by eye bulging. Patients receiving the drug experienced responder rates of approximately 50–54%, compared with just 15% in the placebo group. Improvements were also seen in diplopia (double vision), with responder rates reaching as high as 61% in the more frequent dosing group.
 The treatment was generally well tolerated, with most side effects classified as mild. Notably, no treatment-related serious adverse events were reported, and over 90% of patients completed the full course of therapy.
The treatment was generally well tolerated, with most side effects classified as mild. Notably, no treatment-related serious adverse events were reported, and over 90% of patients completed the full course of therapy.
“We are excited by today’s positive REVEAL 2 results and view these data as a major step forward for the chronic TED patient population. Given the IV-like proptosis response and our plans to launch with an at-home autoinjector, we believe elegrobart can meaningfully attract chronic patients to seek treatment. Elegrobart’s unmatched simplicity and convenience could uniquely drive expansion of the large and underserved chronic TED market,” said Steve Mahoney, President and Chief Executive Officer of Viridian Therapeutics. “With our anticipated launch of veligrotug, which is a short IV infusion course, and two positive phase 3 REVEAL pivotal clinical trials supporting both Q4 weekly and Q8 weekly subcutaneous dosing for elegrobart, our portfolio has the potential to offer anti-IGF-1R efficacy and safety in convenient treatment regimens for TED patients with active or chronic disease.”
Viridian said it plans to submit a biologics license application (BLA) to the FDA in the first quarter of 2027. The company is also preparing for a separate potential product launch: veligrotug, another TED treatment currently under FDA priority review, with a decision expected by June 30, 2026.
