Key Takeaways
-
MCO-010 optogenetic gene therapy produced sustained, clinically meaningful vision improvements of approximately 3 ETDRS lines over three years in patients with advanced retinitis pigmentosa
-
Greater treatment response was associated with higher retinal transduction, better baseline visual acuity (<2.25 LogMAR), and central subfield thickness >150 µm, while genetic mutation type did not impact efficacy
-
The therapy was well tolerated with no serious adverse events reported
Christine N. Kay, MD, Director of Clinical Research and Retinal Genetics, Vitreoretinal Associates in Gainesville, Florida, presented new post-hoc clinical analyses on Nanoscope Therapeutics' lead optogenetic therapy, MCO-010, at the 2026 Macula Society Annual Meeting, which took place February 25-28, in San Diego.
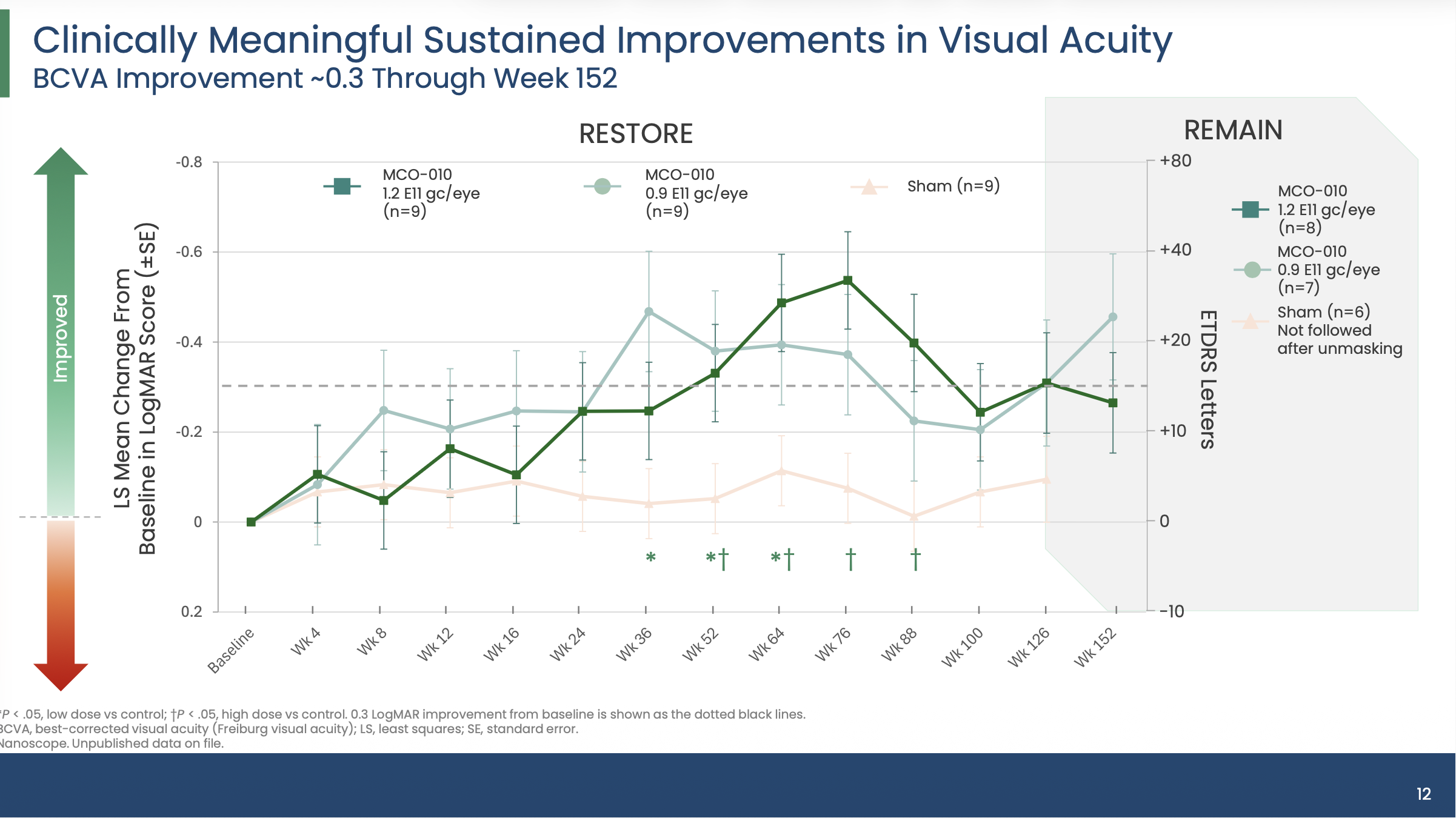
The new data from the Phase 2b/3 RESTORE trial and its long-term follow-up study, REMAIN, revealed that MCO-010 delivered sustained and clinically meaningful vision improvements over 3 years in patients with advanced retinitis pigmentosa (RP).
The therapy is administered as a single intravitreal injection using an AAV2 vector platform and is designed to restore light sensitivity in retinal cells independent of a patient’s specific genetic mutation. Investigators reported that improvements in best-corrected visual acuity (BCVA) were maintained through 152 weeks, with favorable safety findings.
The RESTORE study enrolled 27 patients with advanced RP, randomized to receive a single intravitreal injection of MCO-010 at either 0.9×10¹¹ genome copies (gc)/eye or 1.2×10¹¹ gc/eye, or sham treatment. Patients who received active therapy were eligible to enroll in REMAIN, the long-term extension study, after Week 100.
At Weeks 52 and 76, patients receiving the higher 1.2×10¹¹ gc/eye dose experienced statistically significant improvements in BCVA compared with sham. Mean BCVA improvements were:
-
0.337 ± 0.083 LogMAR at Week 52 (P=0.021 vs sham)
-
0.539 ± 0.103 LogMAR at Week 76 (P=0.001 vs sham)
Patients in the lower-dose 0.9×10¹¹ gc/eye group also showed improvements:
-
0.382 ± 0.124 LogMAR at Week 52 (P=0.029 vs sham)
-
0.374 ± 0.133 LogMAR at Week 76 (P=0.065 vs sham)
By Week 152 in the REMAIN extension study, vision gains remained durable. Mean BCVA improvements from baseline were:
-
0.264 ± 0.112 LogMAR in the 1.2×10¹¹ gc/eye group
-
0.453 ± 0.140 LogMAR in the 0.9×10¹¹ gc/eye group
These improvements correspond to approximately 3 lines on the ETDRS eye chart.
Baseline Vision and Retinal Anatomy Predict Outcomes
Post hoc multivariate analyses examined anatomical and disease-related baseline factors associated with optimal visual outcomes.
Investigators found that:
-
Greater MCO-010 expression, as measured by fundus autofluorescence (FAF) imaging, positively correlated with BCVA gains
-
Patients with baseline visual acuity better than 2.25 LogMAR experienced more favorable outcomes
-
Baseline central subfield thickness (CST) greater than 150 microns was associated with improved treatment response
-
No correlation was observed between specific genetic mutations and efficacy outcomes
The absence of genotype dependence reinforces the mutation-agnostic design of the therapy, potentially broadening its applicability across the heterogeneous RP patient population.
MCO-010 was generally well tolerated over the 3-year follow-up period. The most commonly reported adverse event was anterior inflammation, which was mild to moderate in severity and controlled with topical steroids. Only one patient required ongoing topical steroid use at Week 152. No serious adverse events were reported.
Based on the sustained efficacy and favorable safety profile demonstrated in RESTORE and REMAIN, a rolling Biologics License Application (BLA) submission to the FDA is currently underway.
