Regeneron announced long-term outcomes and new analyses of pivotal clinical data for Eylea HD (aflibercept) Injection 8 mg will be presented at the American Academy of Ophthalmology (AAO) annual meeting in San Francisco from November 3-6.
“Following the approval of Eylea HD, we have continued to evaluate data from the pivotal clinical program to identify new insights that may support physicians in administering this innovative treatment to patients living with certain serious retinal diseases,” Boaz Hirshberg, MD, Senior Vice President, Clinical Development, Internal Medicine at Regeneron, said in a company news release. “At this year’s AAO, we will present a wide-ranging review of results from the PULSAR and PHOTON trials in wet age-related macular degeneration and diabetic macular edema. Together, they showcase the impressive durability and consistent safety profile of EYLEA HD over the long-term across different extended dosing intervals and patient characteristics.”
Notable presentations include a detailed review of positive 2-year (96 weeks) results from the pivotal PULSAR and PHOTON trials investigating Eylea HD with 12- and 16-week dosing regimens, compared to Eylea (aflibercept) Injection 8-week dosing regimens, in patients with wet age-related macular degeneration (AMD) and diabetic macular edema (DME), respectively. Additionally, subgroup data from PHOTON in DME across sex, age, race and ethnicity will be presented, as well as a pooled safety analysis from the PULSAR, PHOTON and phase 2 CANDELA trials.
Two-year data from the pivotal PULSAR trial were previously presented at the Euretina Congress in October 2023. Two-year data from the pivotal PHOTON trial were previously presented at the American Society of Retina Specialists (ASRS) annual meeting in July 2023.
Eylea HD (known as aflibercept 8 mg outside of the U.S.) is being jointly developed by Regeneron and Bayer AG. In the US, Regeneron maintains exclusive rights to Eylea and Eylea HD. Bayer has licensed the exclusive marketing rights outside of the U.S., where the companies share equally the profits from sales of EYLEA and EYLEA HD following any regulatory approvals.
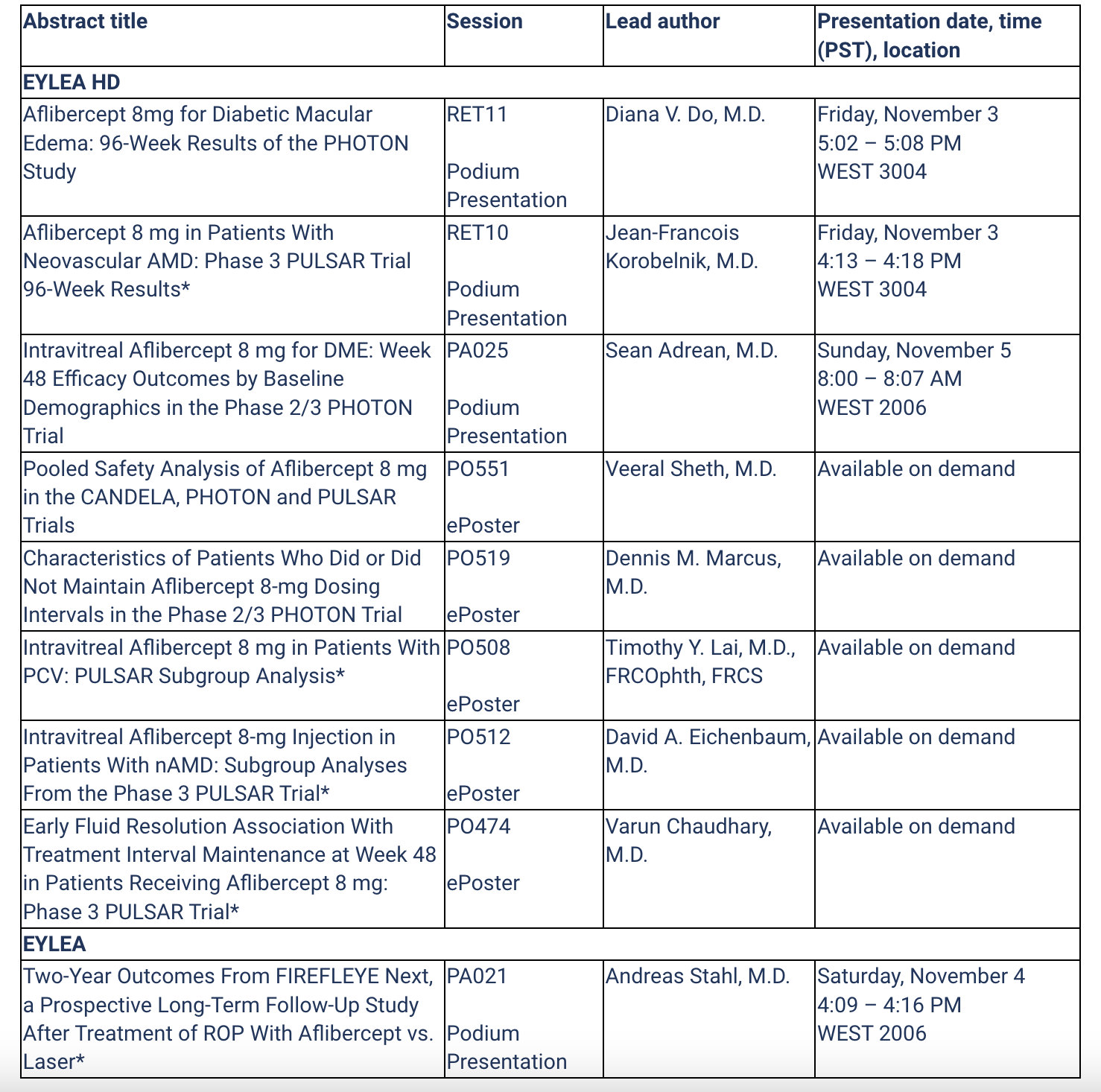
EYLEA HD and EYLEA presentations at AAO:
About the EYLEA HD Clinical Trial Program
PULSAR in wAMD and PHOTON in DME/diabetic retinopathy (DR) are double-masked, active-controlled pivotal trials that are being conducted in multiple centers globally. In both trials, patients were randomized into 3 treatment groups to receive either: EYLEA HD every 12 weeks, EYLEA HD every 16 weeks, or EYLEA every 8 weeks. The lead sponsors of the trials were Bayer for PULSAR and Regeneron for PHOTON.
Patients treated with EYLEA HD in both trials had 3 initial monthly doses, and patients treated with EYLEA received 3 initial doses in PULSAR and 5 in PHOTON. In the first year, patients in the EYLEA HD groups could have their dosing intervals shortened down to an every 8-week interval if protocol-defined criteria for disease progression were observed. Intervals could not be extended until the second year of the study. Patients in all EYLEA groups maintained a fixed 8-week dosing regimen throughout their participation in the trials.
CANDELA was a Regeneron-sponsored Phase 2 trial investigating the safety and efficacy of EYLEA HD extended dosing regimens compared to EYLEA in wAMD patients.
