Key Takeaways
- According to Oculis's DME AWARE Delphi study, a majority of DME patients remain untreated or inadequately treated, highlighting significant gaps in current care and the limitations of invasive therapies
- Findings support strong interest in eye drop therapies like OCS-01 as a potential first-in-class, noninvasive option for early-stage DME and patients with poor response to existing treatments
Oculis announced the presentation of new findings from its DME AWARE Delphi study at the Association for Research in Vision and Ophthalmology (ARVO) 2026 annual meeting.
The DME AWARE initiative, sponsored by Oculis, is a global Delphi study involving a steering committee and 25 leading retina and ophthalmology experts. The initiative used three rounds of anonymous surveys to identify unmet needs in DME management, focusing on disease detection, assessment, and treatment strategies.
Findings from the study confirmed several critical gaps in current care. Experts emphasized the need for noninvasive treatment options, therapies suitable for early intervention, and new treatments that can be used alongside existing standards of care. The panel also established definitions for early disease management, describing “early detection” as identifying DME before fluid accumulation and “early intervention” as treating patients before functional vision decline occurs.
The final survey results further reinforced strong consensus around the need for non-invasive therapies, particularly supporting the use of eye drops as a potential early intervention approach.
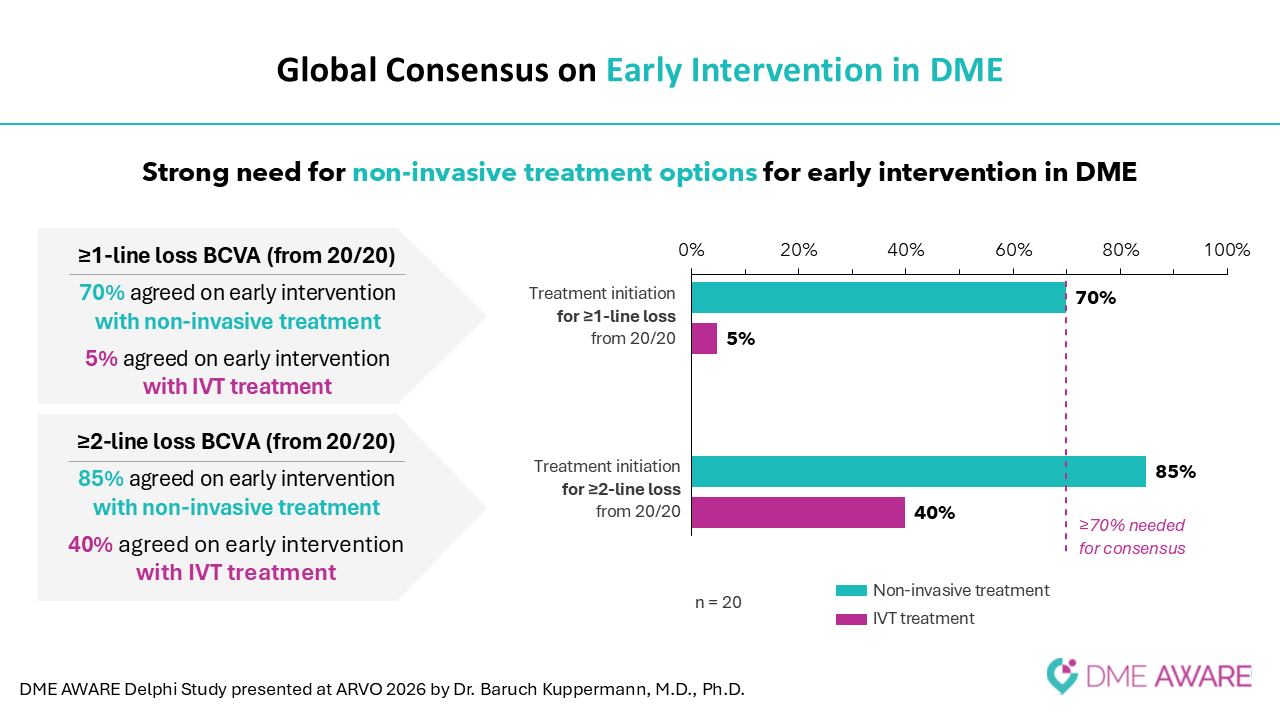
“These findings reflect a truly global expert consensus on the most pressing challenges in DME management,” said Professor Baruch D. Kuppermann, MD, PhD, of the University of California, Irvine, who presented the findings at ARVO. “They underscore the balance between the need for effective disease control and the limitations of current treatment approaches, while pointing toward the promise of less invasive therapies to improve patient outcomes.”
In April, Oculis completed enrollment in both phase 3 DIAMOND-1 and DIAMOND-2 trials of OCS-01 eye drops in DME. The trials are designed as pivotal registration studies to support global marketing applications including NDA submission and approval by the FDA. The trials enrolled over 800 patients at 119 investigative sites throughout the US and several other countries. Topline data from both phase 3 trials is expected in the second quarter of 2026, with NDA submission to follow. If approved, OCS-01 is expected to become the first topical eye drop for the treatment of DME and address unmet medical needs for early treatment intervention or for patients with inadequate response to anti-VEGF therapy.
“With 60% of patients remaining untreated due to the invasive nature of current therapies, there is a clear need for change,” said Oculis CEO Riad Sherif, MD. “As we approach the upcoming readout of the pivotal DIAMOND trials, our data suggest that a topical therapy like OCS-01 could significantly shift the treatment paradigm. It offers a noninvasive option for early intervention and could address the needs of patients who do not respond adequately to injections.”
