Ocular Therapeutix announced positive topline results from its SOL-1 phase 3 superiority trial evaluating Axpaxli (also known as OTX-TKI) for the treatment of wet age-related macular degeneration (AMD).
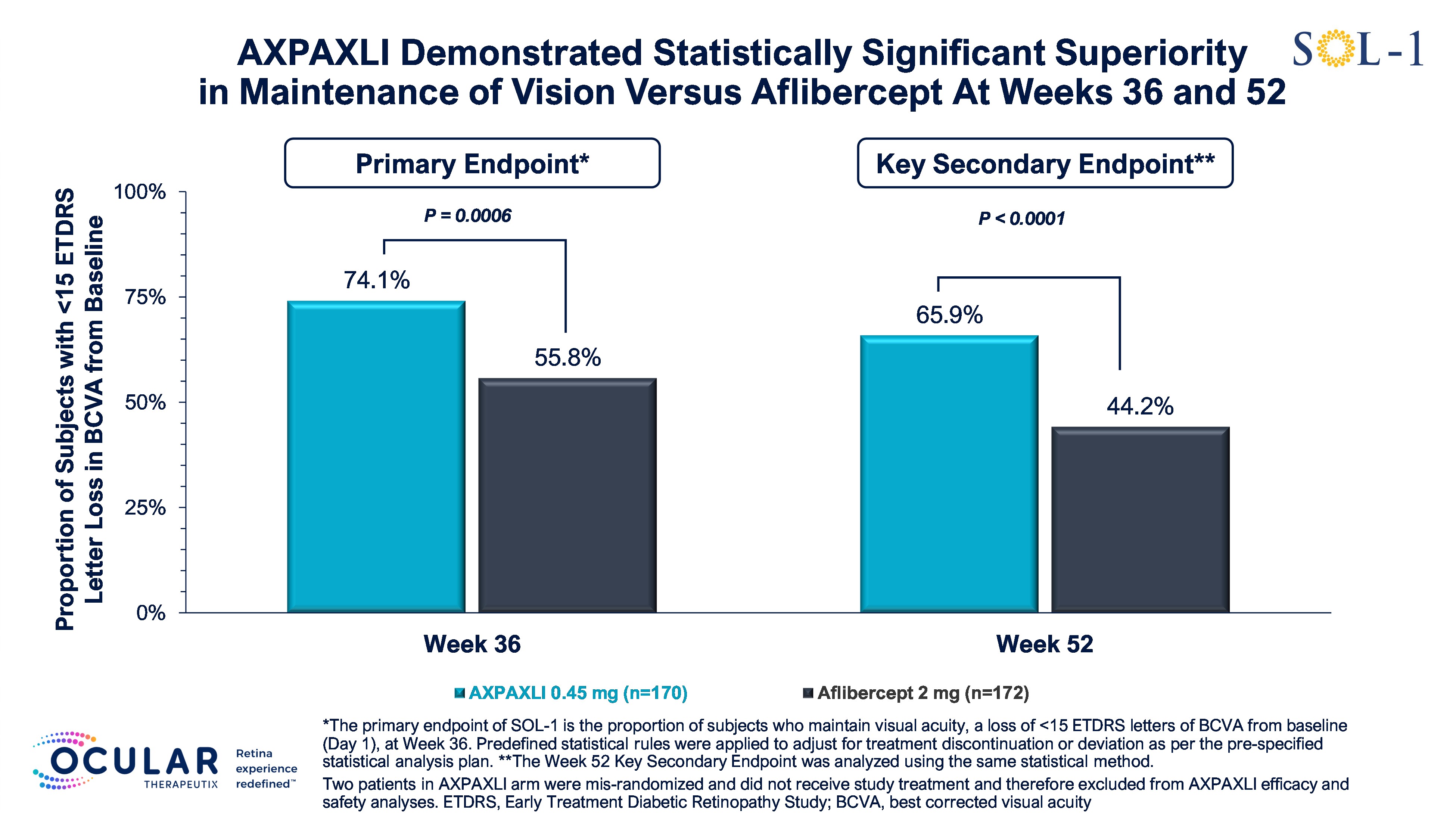
The SOL-1 trial met its primary endpoint with high statistical significance, demonstrating superiority of a single dose of Axpaxli (0.45 mg) compared with a single dose of aflibercept (2 mg) at Week 36. Under the pre-specified statistical analysis plan, aligned with the FDA through a Special Protocol Assessment (SPA), 74.1% of patients treated with Axpaxli maintained visual acuity—defined as a loss of fewer than 15 ETDRS letters—compared with 55.8% of patients treated with aflibercept, representing a risk difference of 17.5% (P=0.0006). Multiple sensitivity analyses confirmed the findings.
In addition to achieving the primary endpoint, Axpaxli demonstrated either statistical significance or numerical superiority across key secondary and prespecified exploratory endpoints, including durability, rescue-free rates, and retinal fluid control.
“We are thrilled to report today’s historic data that position Axpaxli to potentially become one of the most consequential advances in retina,” said Pravin U. Dugel, MD, Executive Chairman, President and CEO of Ocular Therapeutix. “Demonstrating superiority to a single dose of aflibercept under the FDA’s stringent evidentiary standards is an exceptionally high bar. The consistency, durability, and safety profile observed in SOL-1 provide compelling evidence of axitinib’s potency combined with the strength of our proprietary hydrogel platform.”
Durability and Reduced Treatment Burden
Axpaxli met a key secondary endpoint evaluating durability at Week 52. At that timepoint, 65.9% of patients in the Axpaxli arm maintained visual acuity compared with 44.2% in the aflibercept arm, corresponding to a risk difference of 21.1% (P<0.0001). Approximately two-thirds of Axpaxli-treated patients remained rescue-free through Week 52, suggesting the potential for extended dosing intervals and a meaningful reduction in treatment burden, according to Ocular.
Rescue-free rates favored Axpaxli at all assessed timepoints, with 80.6%, 74.7%, and 68.8% of patients remaining rescue-free at Weeks 24, 36, and 52, respectively, compared with 72.1%, 56.4%, and 47.7% for aflibercept.
“Axpaxli is the first and only drug with a novel mechanism to successfully demonstrate superiority to an approved anti-VEGF therapy in an FDA-aligned phase 3 study for wet AMD,” said Arshad M. Khanani, MD, MA, FASRS, Director of Clinical Research at Sierra Eye Associates and Steering Committee Chair for the SOL Program. “The sustained anatomic and visual outcomes, combined with the high proportion of patients remaining rescue-free, suggest Axpaxli could potentially be an annual treatment option for many patients.”
Superior Fluid Control and Favorable Safety Profile
As a pre-specified exploratory endpoint, the trial assessed central subfield thickness (CSFT) stability. At Week 36, 55.9% of Axpaxli-treated patients maintained CSFT within 30 microns of baseline compared with 37.8% in the aflibercept arm (nominal P=0.0013). This superior fluid control was sustained through Week 52.
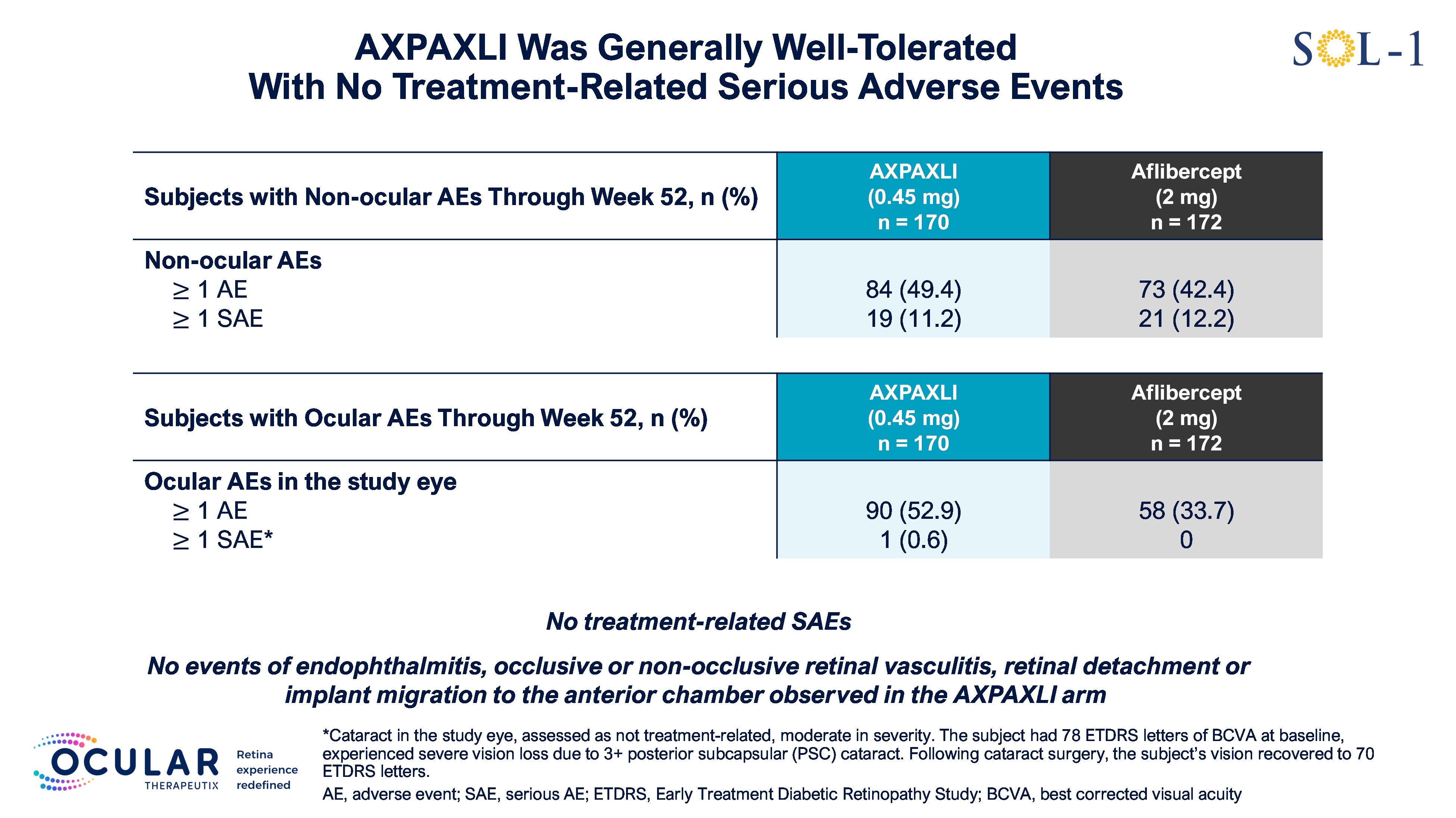
As of the Week 52 database lock on February 5, 2026, Axpaxli was generally well tolerated, with no treatment-related ocular or systemic serious adverse events reported. Notably, there were no cases of endophthalmitis, retinal vasculitis, retinal detachment, or implant migration observed in the Axpaxli arm.
Ocular Therapeutix plans to present detailed SOL-1 results at the 49th Macula Society Annual Meeting later this month. The company intends to pursue discussions with the FDA regarding submission of a new drug application based on the SOL-1 data. If approved, Axpaxli could become the first tyrosine kinase inhibitor commercialized for wet AMD and potentially the only therapy in the indication with a superiority label.
The complementary SOL-R phase 3 noninferiority trial of Axpaxli in wet AMD is ongoing, with topline data expected in the first quarter of 2027.
