A genetics study published this week in The American Journal of Human Genetics reveals that carrying a genetic variant linked to inherited retinal degeneration (IRD) does not guarantee vision loss, challenging long-standing assumptions in human genetics and ophthalmology.
Traditionally, inherited retinal diseases—including conditions such as retinitis pigmentosa and other degenerative retinal disorders—have been considered Mendelian diseases: caused by mutations in single genes that almost always lead to the disease. However, new research led by scientists at Mass General Brigham and Harvard Medical School suggests this dogma may be outdated.
In an extensive analysis using large population biobanks, including the NIH’s All of Us Research Program (AoU) and the UK Biobank, researchers screened nearly 318,000 participants for variants in 33 genes previously associated with IRDs. Among individuals who carried these genetic changes, only about 28% displayed clinical signs of retinal degeneration when broader criteria for disease (including retinal disease and vision loss) were applied.
When stricter clinical codes for IRD were used, the penetrance—the proportion of variant carriers who actually develop disease—dropped to just 9.4%. Even when including possible signs of retinal disease based on imaging, only 16-28% of carriers showed evidence consistent with IRDs in both the AoU and UK Biobank data.
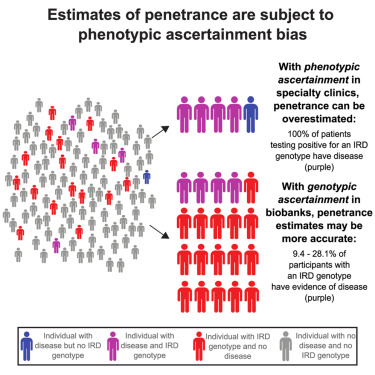
These findings sharply contrast with the traditional view that such genetic variants lead to disease in nearly every carrier. “Our study indicates that the number of people in the general population with genetic variants linked to inherited retinal disorders is much higher than previously thought,” said senior study author Eric Pierce, MD, PhD, director of the Ocular Genomics Institute at Massachusetts Eye and Ear. “Population penetrance of these genes is markedly lower than traditionally assumed.”
The study suggests that additional genetic modifiers or environmental factors likely influence whether a person with a pathogenic IRD gene will develop vision loss. This may have implications for how clinicians interpret genetic test results and counsel patients. Rather than a deterministic “one gene, one disease” model, the new findings support a more nuanced view in which genetics is a significant risk factor but not a certainty.
Researchers say this could influence the future of clinical genetic testing, potentially shifting toward models that account for polygenic and environmental modifiers to more accurately predict outcomes for patients and families. It may also inform future therapeutic strategies by identifying protective factors that prevent disease in unaffected carriers.
