Kodiak Sciences announced follow-up data from the ongoing APEX phase 1b study evaluating KSI-101 in patients with macular edema secondary to inflammation (MESI).
KSI-101 is a novel, high-strength (100 mg/mL) antibody-based investigational therapy featuring a bispecific mechanism of action that simultaneously targets interleukin-6 (IL-6) and vascular endothelial growth factor (VEGF).
APEX Study Week 20 Results
In the APEX phase 1b study, patients were dosed with KSI-101 at Weeks 0, 4, 8, and 12. By Week 20, KSI-101 continued to demonstrate a favorable safety and tolerability profile with no new safety signals observed.
Key highlights include:
-
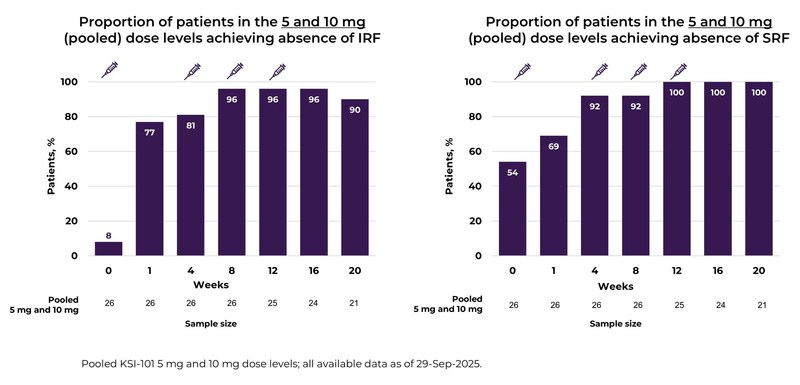
Sustained retinal drying through Week 20, with a majority of patients achieving complete resolution of intraretinal and subretinal fluid
-
Meaningful visual acuity improvements, including normalization to 20/25 Snellen equivalent in many patients
-
Rapid onset of action, with most drying effect observed as early as Week 1
“These Week 20 follow-up results suggest even more strongly the potential for KSI-101 to become a cornerstone therapy for the treatment of MESI,” said Victor Perlroth, MD, Chairman and Chief Executive Officer of Kodiak Sciences. “The data show that achieving complete retinal drying and meaningful visual acuity gains is possible for most MESI patients—with a favorable safety profile. If confirmed in our ongoing phase 3 studies, PEAK and PINNACLE, KSI-101 may harmonize the MESI therapeutic landscape as a safe, first-line unifying therapy for all causes of MESI.”
“At the recent American Academy of Ophthalmology meetings, IL-6 inhibition was shown in phase 3 studies to deliver significant improvements in vision and anatomy for uveitic macular edema—a key subset of MESI. KSI-101, by combining potent IL-6 and VEGF inhibition, may offer even greater clinical benefit and achieve best-in-class efficacy," Dr. Perlroth added.
“The extended Week 20 data further validate KSI-101’s transformative potential. We observed real, sustained retinal dryness in over 90% of patients and near-normal visual acuity of 20/25 Snellen equivalent," said Velazquez-Martin, MD, Chief Medical Officer of Kodiak Sciences. "The rapid onset of effect across all dose levels, with most drying seen by Week 1, is a remarkable differentiator. These data strongly support advancing the 5 mg and 10 mg doses into our pivotal phase 3 program.”
The Phase 3 PEAK and PINNACLE studies—evaluating the 5 mg and 10 mg doses of KSI-101—are actively enrolling patients with macular edema secondary to inflammation. Final APEX results, including Week 24 data, are expected to be presented by Sumit Sharma, M.D., retina and uveitis specialist at the Cleveland Clinic’s Cole Eye Institute, on February 7, 2026, at the Angiogenesis, Exudation, and Degeneration 2026 Annual Meeting.
Week 20 follow-up results from the APEX study are available on Kodiak’s website at: https://ir.kodiak.com/events-and-presentations/events
