Apellis Pharmaceuticals announced new findings from a post hoc analysis of the GALE extension study, revealing that continuous treatment with Syfovre (pegcetacoplan) over 5 years meaningfully slowed the progression of geographic atrophy (GA) secondary to age-related macular degeneration (AMD).
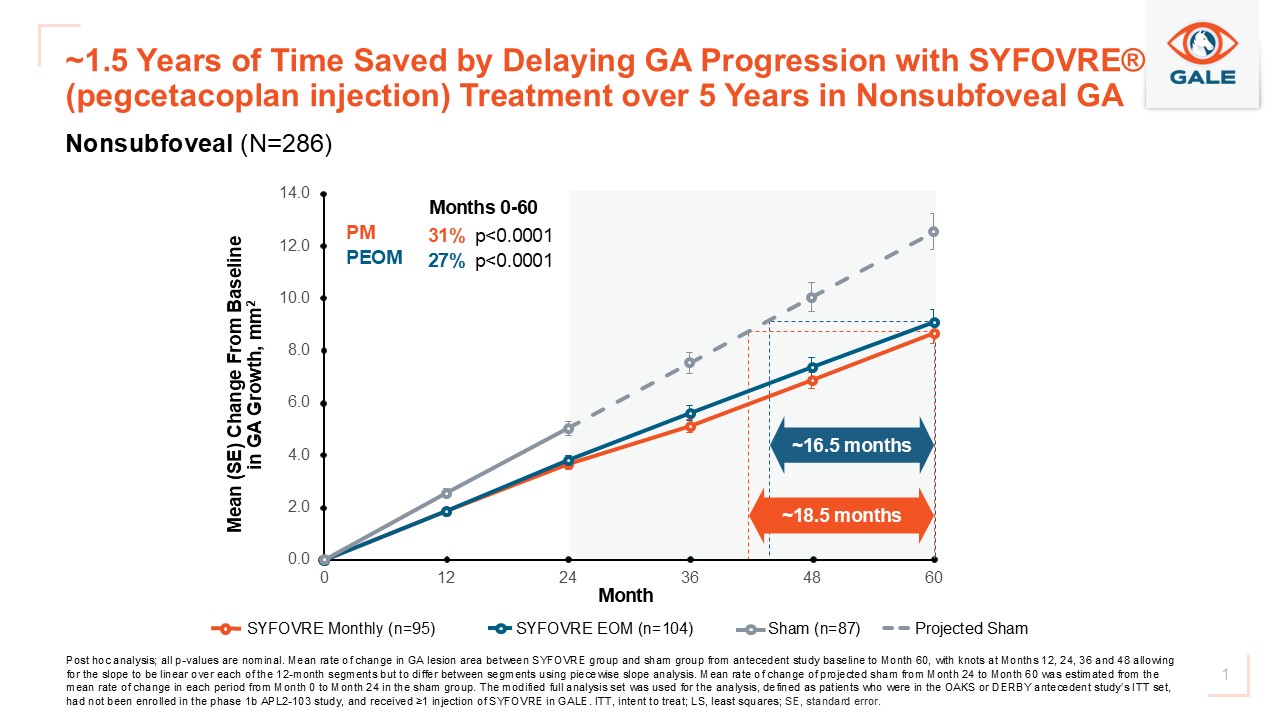
According to the analysis, both every-other-month and monthly dosing regimens of Syfovre delayed GA lesion growth by approximately 1.5 years in patients with nonsubfoveal GA compared with sham or projected sham treatment.
“I’m very encouraged by these long-term results, which show that early and continuous treatment with Syfovre can meaningfully delay the progression of GA,” said Dilsher Dhoot, MD, of California Retina Consultants. “Importantly, these data indicate that Syfovre alters the natural course of this disease, which causes irreversible vision loss and profoundly impacts patients’ daily lives.”
“These 5-year results underscore the transformative and durable impact of targeting C3 with Syfovre to delay the progression of GA,” added Caroline Baumal, MD, chief medical officer at Apellis. “With the most extensive data set in GA, our broad clinical and real-world experience has greatly advanced the retina community’s understanding of this devastating disease and reinforced Apellis’ leadership.”
Apellis reported that the safety profile of Syfovre through 5 years remained consistent with previously published data, with no new safety signals observed. The company plans to present detailed results at an upcoming medical meeting.
About the GALE Long-Term Extension Study
The GALE study (n=792) is a phase 3, multicenter, open-label extension study evaluating the long-term efficacy and safety of Syfovre in patients with geographic atrophy secondary to AMD. More than 80% of participants from the pivotal OAKS and DERBY trials continued into GALE.
GALE’s objectives include assessing the long-term incidence and severity of ocular and systemic treatment-emergent adverse events, as well as changes in total GA lesion area, measured by fundus autofluorescence imaging.
Patients included in the 5-year lesion growth analysis had received Syfovre treatment through Month 24 in the OAKS and DERBY studies and continued on the same regimen in GALE. Because sham-treated patients transitioned to Syfovre after Month 24, researchers used a projected sham arm—validated by a fellow eye analysis—to model untreated disease progression from Months 24 to 60. This projection was based on the observed linear lesion growth rate from the OAKS and DERBY sham arms over the initial 2 years.
